HIGH-RESOLUTION CARTOGRAPHY OF THE TRANSCRIPTOME AND METHYLOME LANDSCAPES OF DIFFUSE GLIOMAS
A high degree of molecular heterogeneity is a fundamental characteristic of diffuse gliomas, a brain tumor entity, which splits into several subtypes of different but overall adverse prognosis. Heterogeneity is governed by a handful of key mutations—first of all, of the isocitrate dehydrogenase gene. It drastically affects DNA methylation on a genome-wide scale. DNA methylation acts as an important regulator of gene transcription with consequences for glioma physiology. We here present a combined gene expression and DNA methylation study with the focus on lower-grade (II–III), adult-type gliomas. It aimed at deciphering glioma heterogeneity into molecular subtypes at a finer granularity level and at characterizing the underlying modes of gene regulation. Our analysis made use of high-resolution molecular portrayal, a machine learning approach to visualize complex genomic data. The results support the importance of epigenetics for glioma diversity and, in consequence, for prognosis and epigenetics-directed treatment.
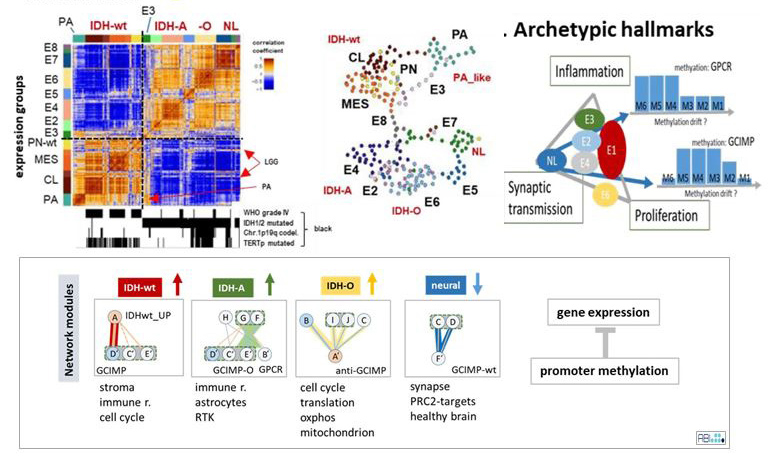
Molecular mechanisms of lower-grade (II–III) diffuse gliomas (LGG) are still poorlyunderstood, mainly because of their heterogeneity. They split into astrocytoma- (IDH-A) andoligodendroglioma-like (IDH-O) tumors both carrying mutations(s) at the isocitrate dehydrogenase(IDH) gene and into IDH wild type (IDH-wt) gliomas of glioblastoma resemblance. We generateddetailed maps of the transcriptomes and DNA methylomes, revealing that cell functions dividedinto three major archetypic hallmarks: (i) increased proliferation in IDH-wt and, to a lesser degree,IDH-O; (ii) increased inflammation in IDH-A and IDH-wt; and (iii) the loss of synaptic transmis-sion in all subtypes. Immunogenic properties of IDH-A are diverse, partly resembling signaturesobserved in grade IV mesenchymal glioblastomas or in grade I pilocytic astrocytomas. We analyzeddetails of coregulation between gene expression and DNA methylation and of the immunogenicmicro-environment presumably driving tumor development and treatment resistance. Our transcrip-tome and methylome maps support personalized, case-by-case views to decipher the heterogeneityof glioma states in terms of data portraits. Thereby, molecular cartography provides a graphicalcoordinate system that links gene-level information with glioma subtypes, their phenotypes, andclinical context.
click here for reading the full article https://www.mdpi.com/2072-6694/13/13/3198
